 The replacement of paper with automation and AI is a seminal, defining change for how life science companies manage quality and compliance. New appendix about modern infrastructure and infrastructure management A new section has been added to clarify these new expectations.ġ0. New appendix about use of agile within a GxP environmentĪgility is a core ingredient of the Second Edition. New appendix about AI and machine learningĩ. New appendix about blockchain and distributed ledger technologyĨ. 
The appendix also clarifies new expectations around electronic records, signatures and audit trails.ħ. The rise of cloud based technology and block chain has been taken into account. Appendix on electronic production of records updated  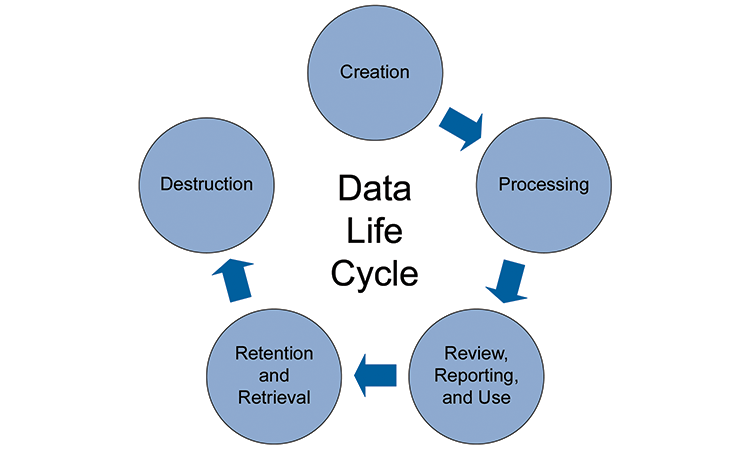
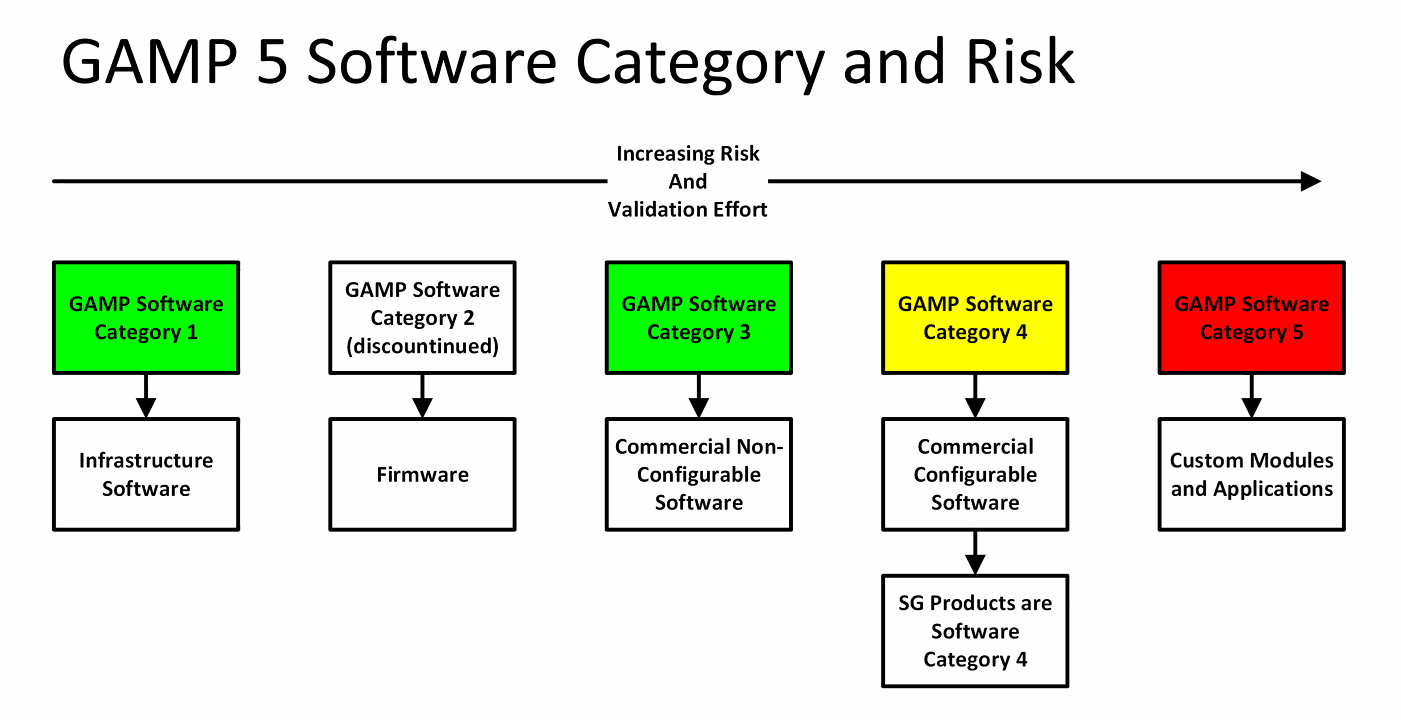
The areas of GAMP 5 focusing on user requirements specifications (URS) and functional/design specifications have been tweaked to reflect the new world of modern, #agilesoftware.ĥ. There's therefore a shift in emphasis from traditional documents like IQs, OQs and PQs to risk-based records of information, held in appropriate systems, that take into account the modern software lifecycle.Ĭritical thinking is a huge cornerstone of computerized system assurance, both in the GAMP 5 Second Edition and the FDA's new guidelines unveiled in September 2022. Legacy GAMP validation documents which assume a linear software approach, like installation, operation and performance qualification documents, are no longer relevant for the Second Edition. The Second Edition recognizes the largely non linear, agile and more cyclical nature of modern software development. The First Edition of GAMP 5 assumed a broadly linear approach to software development, which no longer reflects how modern software is brought to life. In keeping with the broad emphasis shift to agile, risk based adoption of modern digital tools for GxP-regulated businesses, the Second Edition has brought 10 key changes to how 'good automated manufacturing practice' is defined. It should be seen as part of a broader industry trend taking place: the evolution from computerized system validation (CSV) to computerized system assurance (CSA). The GAMP 5 2nd Edition, was released in July 2022, replacing the First Edition unveiled in 2008. There are five key concept of this guide –Ĭategory 1: Infrastructure software (example: operating system, database engines, programming etc.)Ĭategory 2: No longer available in GAMP 5Ĭategory 3: Non-Configured products (example: firmware based applications)Ĭategory 4: configured products (example: LIMS, ERP, Spread sheets etc.)Ĭategory 5: Custom applications (example: varies but include internally or externally developed IT applications) These are collectively known as GxP regulations. This guide applies to computerized systems used in the regulated activities covered by: GAMP guidance has been designed for use by a wide range of disciplines and responsibilities, including:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
